 Thus, when silver nitrate and an alkali metal hydroxide compound react, the formation of Ag 2 O takes place. Now slowly separate the silver nitrate solution into the beaker and keep the remaining silver oxide of the test tube in a sealed container as it is a toxic compound and dry it. Keep the test tube undisturbed for 20 minutes, so that the precipitate will settle down at the bottom. As both the reagents mix well, a dark brown or black coloured precipitate forms which indicates the formation of silver oxide. Now pipette out 20ml of sodium hydroxide in the same test tube. To prepare silver oxide from silver nitrate, we take 20ml of silver nitrate in a clean, dry test tube. Alkali hydroxide is those compounds that are composed of an alkali metal cation and hydroxide anion, for example, LiOH, NaOH, KOH, etc. An aqueous solution of silver nitrate is prepared, and it is mixed with a solution of alkali hydroxide. Silver oxide can be prepared by using another silver compound which is silver nitrate. The structure of its unit cell is given below – The space group includes a point group with full octahedral symmetry. A quarter of the body diagonal shifts its one sublattice. In its unit cell Ag atoms arrange in the face centred cubic sublattice and oxygen atoms in the base centred cubic sublattice. It has two coordinate silver centres linked by tetrahedral oxides. Silver oxide has the same structure as the Cu 2 O molecule. The silver oxide that is both Ag 2 O and AgO work as a cathodic material (Control corrosion of a metal surface) in silver-zinc rechargeable batteries. This powder is used to make other silver compounds. The colour of silver oxide is black or dark brown. The chemical compound of silver oxide with the formula is Ag 2 O. It is also known as silver dust, argentous oxide, and silver monoxide. It is mainly used in batteries and in the preparation of other silver compounds. It is a black or dark brown coloured compound. In silver(I) oxide, (I) shows here the oxidation number of silver which is +1. The molecular formula of silver oxide is Ag 2 O. Silver oxide is an inorganic chemical compound composed of two atoms of silver and one atom of oxygen. This site explains how to find molar mass.In the study of Chemistry, the term oxides describe the reaction of metal or any element with oxygen atoms that results in Metal oxides. The reason is that the molar mass of the substance affects the conversion. 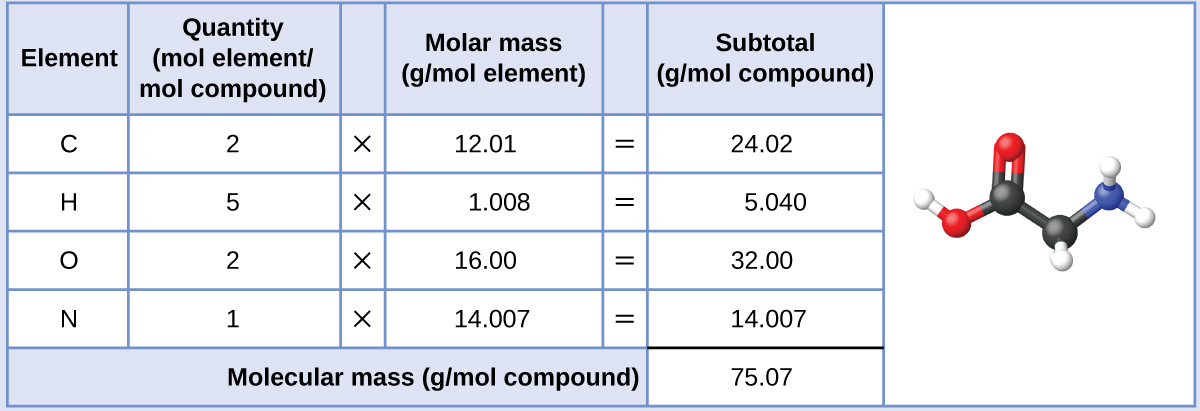
To complete this calculation, you have to know what substance you are trying to convert. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.Ī common request on this site is to convert grams to moles. 
This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. 
The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. 
The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. These relative weights computed from the chemical equation are sometimes called equation weights.įinding molar mass starts with units of grams per mole (g/mol). The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
